Mutations
in DNA are what make each human unique however some of those mutations cause
different life threatening situations commonly called cancer. When someone is
diagnosed with cancer, their cells are mutated in a deadly way and duplicate
quickly so that their body can’t identify what is happening fast enough.
6-Thiopurine (6TP) is an anti-cancer chemotherapy drug used mainly for a
childhood leukemia called acute lymphoblastic leukemia (ALL) as well as other
autoimmune disorders (Malone, n.d.). This chemotherapy drug is used to try to
suppress immune system to a point in which the immune system does not notice
the changes 6-thiopurine has on DNA and RNA in severe cases where use of the
drug is needed. When 6-thiopurine enters the body, it changes from 6-thiopurine
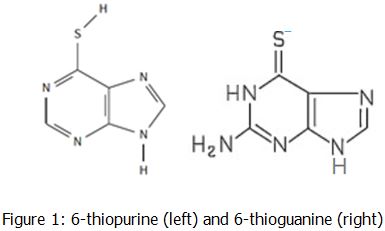
to 6-thioguanine (6TG). Shown in Figure 1 is the change between 6-thiopurine
and 6-thioguanine.
When 6-thiopurine enters the body, it changes into 6-thioguanine in order to act as a the guanine nucleotide in DNA. It can be used in the curing of acute lymphoblastic leukemia (ALL), as an anti-inflammatory, or as an immunosuppressant. When the patient’s other replication enzymes goes to check the DNA for any errors, it does not notice that it is 6-thioguanine instead of a guanine which can cause major problems quickly (Nagai, Nagasawa, Kihara, Okuda & Fujimoto, 2006).
Even though 6-thiopurine is effective on many people, the 50mg dosage given to most people always leaves behind a tiny concentration after some of it is collected by the patient’s metabolism (Malone, n.d.). These small concentrations that are leftover can cause serious side effects such as a dearth in red and white blood cells and any level of liver damage. To prevent any serious damages from occurring in a patient’s body, regular blood monitoring is required. However, the current methods used to detect 6-thiopurine are not competent for use in detecting small quantities (~1 picomole) of 6-thiopurine leaving patients in more pain and doctors without the understanding of why the patients have these side effects. The problem is the detection method. Currently, there are several ways to detect large concentrations of 6-thiopurine such as high pressure liquid chromatography (HPLC) but none, however, can detect very tiny concentrations (Sparrow, n.d.). That is when a method called fluorescence spectroscopy comes into play.
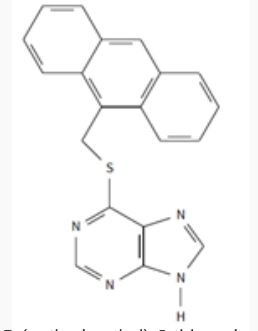
Fluorescence spectroscopy is a specific type of electromagnetic spectroscopy that is used to detect the fluorescence from a sample by shining a light through a quartz cuvette containing the solution (Lakowicz, 2006). This method of detection is the best detection method to date but in order to detect these small amounts, the sample must go through chromatography for separation and purification as well as Nuclear Magnetic Resonance Spectroscopy (NMR) and Infrared Spectroscopy (IR) to find out the molecular structure of the sample. Finally, the sample will go through fluorescence spectroscopy to detect even a small concentration of 6-thiopurine in the sample. However, 6-thiopurine can not actually be detected by fluorescence spectroscopy. Therefore, 6-thiopurine is reacted with 9-(chloromethyl)anthracene S-(anthrylmethyl)-6-thiopurine. When the Fluorescence Spectrophotometer finds the limit of detection (LOD), it is actually detecting the 9-(chloromethyl)-anthracene. SInce there will be the same number of 6-thiopurine as 9-(chloromethyl)-anthracene (shown in Figure 2), the concentration of 6-thiopurine can also be detected. It is hypothesized that fluorescence spectroscopy will be a more effective way to detect small concentrations of 6-thiopurine because it is a more sensitive method of detection than high pressure liquid chromatography (Lakowicz, 2006).
Even though 6-thiopurine is effective on many people, the 50mg dosage given to most people always leaves behind a tiny concentration after some of it is collected by the patient’s metabolism (Malone, n.d.). These small concentrations that are leftover can cause serious side effects such as a dearth in red and white blood cells and any level of liver damage. To prevent any serious damages from occurring in a patient’s body, regular blood monitoring is required. However, the current methods used to detect 6-thiopurine are not competent for use in detecting small quantities (~1 picomole) of 6-thiopurine leaving patients in more pain and doctors without the understanding of why the patients have these side effects. The problem is the detection method. Currently, there are several ways to detect large concentrations of 6-thiopurine such as high pressure liquid chromatography (HPLC) but none, however, can detect very tiny concentrations (Sparrow, n.d.). That is when a method called fluorescence spectroscopy comes into play.
Fluorescence spectroscopy is a specific type of electromagnetic spectroscopy that is used to detect the fluorescence from a sample by shining a light through a quartz cuvette containing the solution (Lakowicz, 2006). This method of detection is the best detection method to date but in order to detect these small amounts, the sample must go through chromatography for separation and purification as well as Nuclear Magnetic Resonance Spectroscopy (NMR) and Infrared Spectroscopy (IR) to find out the molecular structure of the sample. Finally, the sample will go through fluorescence spectroscopy to detect even a small concentration of 6-thiopurine in the sample. However, 6-thiopurine can not actually be detected by fluorescence spectroscopy. Therefore, 6-thiopurine is reacted with 9-(chloromethyl)anthracene S-(anthrylmethyl)-6-thiopurine. When the Fluorescence Spectrophotometer finds the limit of detection (LOD), it is actually detecting the 9-(chloromethyl)-anthracene. SInce there will be the same number of 6-thiopurine as 9-(chloromethyl)-anthracene (shown in Figure 2), the concentration of 6-thiopurine can also be detected. It is hypothesized that fluorescence spectroscopy will be a more effective way to detect small concentrations of 6-thiopurine because it is a more sensitive method of detection than high pressure liquid chromatography (Lakowicz, 2006).
Figure 2: S-(anthrymethyl)-6-thiopurine (the name for reacted 6TP and 9A)